Should be converted to less soluble forms and sent to disposal facilities. 
In closed bottles, air-tight for the anhydrous form. 
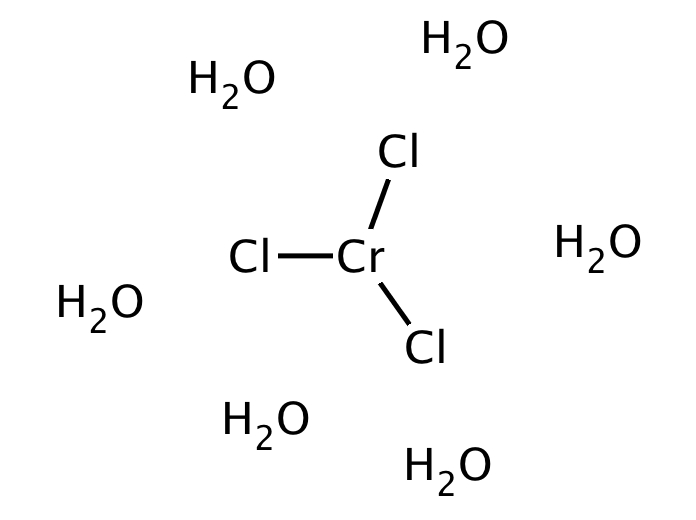
While less toxic than Cr(VI) compounds, chromium chloride is less toxic, although it's still quite harmful. Make Cr coordination complexes and adducts.To obtain the anhydrous form from the hexahydrate, you must heat the CrCl 3 in the presence of a chlorine or chloride source, like thionyl chloride or dry hydrogen chloride gas. The absorption spectrum published shows minima at 480 n m (blue) for the hexaqua and 510 n m (green) for the monochloro. The metabolism of chromic chloride hexahydrate in guinea pigs for 60 days after intratracheal injection of 200 ug Cr 10 min after injection, 69 of the dose remained in the lungs, & only 4 was found in the blood & liver, kidneys, spleen. According Bjerrum and Bjerrum 1 a 1 M chromium (III) chloride solution in equilibrium at 25 C contains about 13 monochloro and 85 hexaqua ions. Heating chromium(III) chloride hexahydrate in air will cause some of it to oxidize, resulting in CrCl 3 contaminated with chromium oxide and oxychloride. The color of the solution may vary as long it is not in equilibrium. 
Reacting chlorine gas with hot chromium metal will also give anhydrous CrCl 3. Chromium (III) chloride tetrahydrofuran complex (1:3) Empirical Formula (Hill Notation): C12H24Cl3CrO3. Alternatively, hexahydrate chromium chloride is treated with thionyl chloride, yielding very dry CrCl 3.Ī more energetic route involves the carbothermic chlorination of chromium(III) oxide between 650–800 ☌. hydrochloric acid.Ĭhromium chloride anhydrous can be prepared by reacting chromium hydroxide, chromium(III) oxide or just chromium metal with hydrochloric acid in methanol. It will be packed in vacuum packing in carton or iron drum.Chromium(III) chloride will react with bases to give chromium hydroxide.Ĭhromium(III) chloride is a purple (anhydrous) or dark green (hydrated) solid.Ĭhromium chloride is sold by chemical suppliers.Ĭhromium chloride hexahydrate can be obtained by reacting chromium oxide, hydroxide or plain chromium metal with conc. It will be carefully handled during storage and transportation to preserve the quality of our product in its original condition. Our Chromium(III) Chloride Hexahydrate Powder is stored in a cool, ventilated warehouse and will be kept sealed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
